



Top Asphalt Contractor Located in Tinton Falls NJ
Full Service Pavement Solution Provider Serving New Jersey Statewide
Garden State Pavement Solutions provides innovative solutions and best practices for cost-effective pavement management.
Garden State Sealing has been a trusted name and partner to the property management community since 1983. In September of 2022, we became Garden State Pavement Solutions. Our new name more accurately depicts who we have become and better emphasizes the strength of our core capabilities.
Our advanced, in-depth property evaluation tools, pavement rating system, and project charter methodology will help property managers effectively budget and plan for current and annual new construction, reconstruction and maintenance. We believe it is important to build trust with our customers through transparency and deed. We A.I.M. for good. Proudly serving Edison, Piscataway, South Brunswick, Princeton, East Windsor, Bridgewater, Cranbury, Woodbridge, Monroe Township, Carteret, Linden and the surrounding communities.
SERVICES WE OFFER
PARKING LOT PAVEMENT
ASSESSMENT & PLANNING
For New Jersey parking lots, Garden State Pavement Solutions is the commercial paving company trusted by more businesses than other paving contractors.
LEARN MORE
INDUSTRIES WE SERVE
COMMERCIAL
INDUSTRIAL
RETAIL
HOA
STATE CONTRACTOR
SCHOOLS & UNIVERSITIES
HOSPITALITY
HEALTH CARE
MUNICIPALITIES
OUR MISSION
Is to be the best in class
at servicing our customers through knowledgeable, accountable teams who hold the company’s and customers’ best interests at their core. We value integrity and trust in our staff, vendors, and customers.
WHY CHOOSE US
Full Service Pavement Solution Provider
We flawlessly execute your services and maintain them into the future to preserve and extend your hard assets.
OUR CULTURE
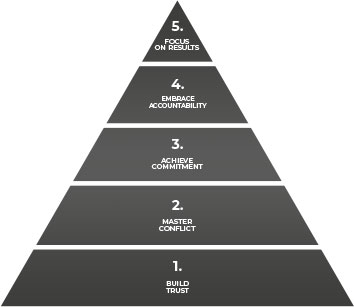
- FOCUS ON RESULTS
- EMBRACE ACCOUNTABILITY
- ACHIEVE COMMITMENT
- MASTER CONFLICT
- BUILD TRUST


























